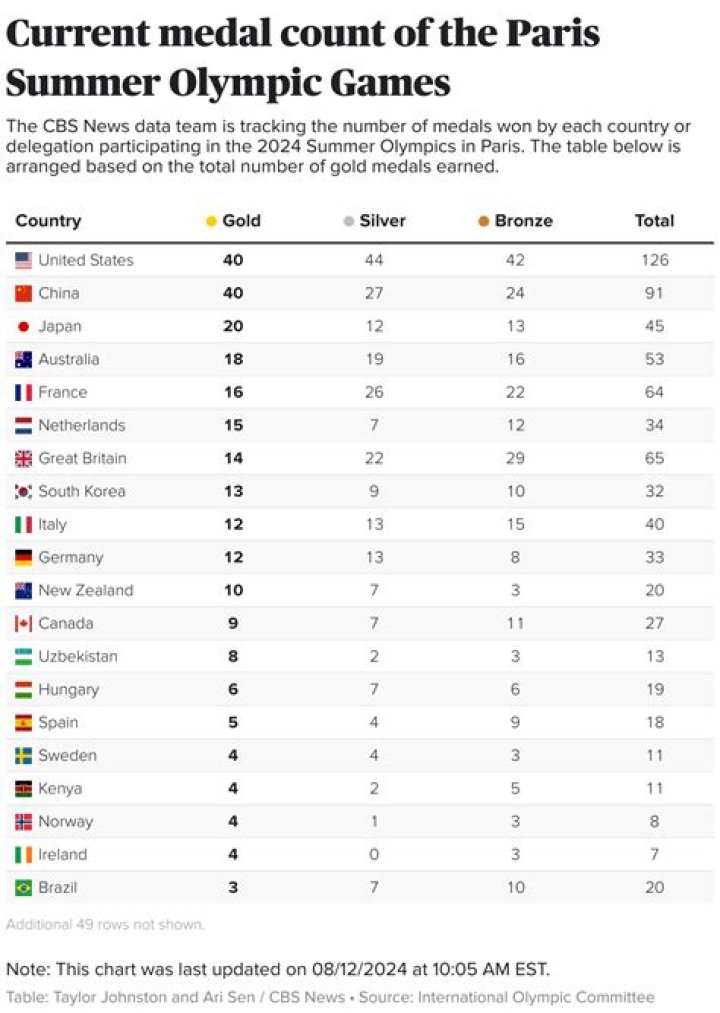
Why is calcium nitrate a salt

John Parsons
Updated on March 31, 2026
Calcium nitrite represented by the chemical formula Ca(NO2)2 or CaN2O4 that bears the IUPAC name calcium dinitride is an odorless white or light yellowish deliquescent powder that is very soluble in water and slightly in alcohol [4, 5]. It is a calcium salt and an ionic compound.
Is calcium nitrite A salt?
Calcium nitrite represented by the chemical formula Ca(NO2)2 or CaN2O4 that bears the IUPAC name calcium dinitride is an odorless white or light yellowish deliquescent powder that is very soluble in water and slightly in alcohol [4, 5]. It is a calcium salt and an ionic compound.
Is calcium nitrate a soluble salt?
Figure 1: Solubility of calcium nitrate in water. … With its relatively high solubility in water it is a highly soluble salt.
Is calcium nitrate a metal salt?
Calcium nitrate, also known as calcium saltpeter or anhydrous calcium nitric acid, is a member of the class of compounds known as alkaline earth metal nitrates. … A variety of related salts are known including calcium ammonium nitrate decahydrate and calcium potassium nitrate decahydrate .What is the nature of calcium nitrate salt?
Answer: Answer:Calcium Nitrate is a highly water soluble crystalline Calcium source for uses compatible with nitrates and lower (acidic) pH. All metallic nitrates are inorganic salts of a given metal cation and the nitrate anion.
Is Calcium a nitrite?
IdentifiersChemical formulaCa(NO 2) 2Density2.26 g/cm3Melting point390 °C (734 °F; 663 K)Hazards
Is calcium nitrate basic or acidic?
NamesUNIINF52F38N1N O8T8H4NBBR (tetrahydrate)UN number1454CompTox Dashboard ( EPA )DTXSID1039719show InChI
What metals react with calcium nitrate?
► Mixtures of Calcium Nitrate with ALKYL ESTERS; PHOSPHORUS; TIN CHLORIDE; and REDUCING AGENTS (such as LITHIUM, SODIUM, ALUMINUM and their HYDRIDES) may result in fires and explosions. (such as HYDROCHLORIC, SULFURIC and NITRIC); METAL SALTS; and COMBUSTIBLES.What happen when calcium nitrate is heated?
Calcium nitrate, on heating, decomposes to give calcium oxide.
Is calcium a metal?Calcium is a silvery-white, soft metal that tarnishes rapidly in air and reacts with water. Calcium metal is used as a reducing agent in preparing other metals such as thorium and uranium. It is also used as an alloying agent for aluminium, beryllium, copper, lead and magnesium alloys.
Article first time published onWhy is calcium nitrate soluble?
Calcium nitrate is a compound that is highly soluble in water. When calcium nitrate dissolves in water, the heat is absorbed. This absorption of heat makes it easily dissolved in water.
Does calcium nitrate cause hardness?
Temporary hardness of water is due to the presence of the nitrates of Ca and Mg.
What is the composition of calcium nitrate?
So, in Calcium nitrate – Calcium is 26.6%, Nitrogen is 9.33% and Oxygen is 64%. and is granular solid which highly absorbs moisture from air. It is widely used as an oxidising agent and as a fertilizer in agriculture. Calcium nitrate is also useful in making explosives and fireworks.
Is calcium nitrate a base or salt?
Calcium as an ion is 2+, while nitrate is 1-. Thus, 2 – 1 – 1 = 0, or a neutral molecule. Yes, calcium nitrate is a salt. Salts are simply metals with non-metals.
Is calcium nitrate ionic or covalent?
The name of the compound Ca(NO3)2 is Calcium nitrate. An ionic compound, is a compound that contains a metal element in its formula unit.
Is cano32 basic?
It is a neutral salt because it is salt of strong Acid and strong Base.
What does calcium nitrate do to plants?
What does calcium nitrate do? It helps with cell formation but it also neutralizes acids to detoxify the plant. The nitrogen component is also responsible for fueling protein production and essentially leafy growth. Heat and moisture stress can cause calcium deficiencies in certain crops, like tomatoes.
Is calcium nitrate a gas?
Calcium nitrate appears as white to light gray granular solid. May be either the anhydrous compound or the tetrahydrate. Used in fertilizers, explosives and pyrotechnics.
Is calcium a bicarbonate?
NamesRelated compoundsOther cationsMagnesium bicarbonate
Is calcium nitrate an electrolyte?
Calcium nitrate is a strong electrolyte. Ca(NO3)2 completely dissociates in an aqueous solution to form Ca2+ cations and NO3- anions; classifying it as a strong electrolyte.
What is the cation for calcium nitrite?
Calcium nitrite | Ca(NO2)2 – PubChem.
When calcium nitrate is heated name the gas evolved?
Note: There is a reddish brown fumes which is released which is the nitrogen dioxide gas.
When calcium nitrate is heated it gives calcium oxide?
When calcium nitrate is heated it gives calcium oxide Nitrogen dioxide gas and Oxygen gas.
Is calcium oxide a gas?
Calcium oxide (CaO), commonly known as quicklime or burnt lime, is a widely used chemical compound. It is a white, caustic, alkaline, crystalline solid at room temperature.
When a piece of calcium reacts with nitric acid What will produce salt?
Calcium oxide reacts with nitric acid (HNO_3) to form aqueous calcium nitrate and water.
Is calcium nitrate harmful to animals?
lethal dose is lower in starving animals and in animals fed a poor diet. sufficient amounts of nitrate are ingested the animals become cyanotic, the pulse is rapid, and the respiration is shortened and frequent and tremor often occurs in some muscle groups.
What salt is formed when calcium reacts with dilute nitric acid?
Additional step: identify the salt. The salt formed is calcium nitrate. Hydrocyanic acid (HCN(aq)) can be neutralized by potassium hydroxide (KOH(aq)). Write a balanced chemical equation for the reaction between these two compounds and identify the salt that it produces.
Why calcium is a metal?
calcium is a metal because it is in group 2 which makes it an alkaline earth metal and a powerful reducing agent that loves to react with nonmetals especially powerful oxidizing agents like fluorine and chlorine.
Is Salt an element?
Chemically, table salt consists of two elements, sodium (Na) and chloride (Cl). Neither element occurs separately and free in nature, but are found bound together as the compound sodium chloride.
Is calcium a solid liquid or gas?
Calcium is a chemical element with symbol Ca and atomic number 20. Classified as an alkaline earth metal, Calcium is a solid at room temperature.
Is calcium nitrate soluble or insoluble?
Calcium Nitrate is a highly water soluble crystalline Calcium source for uses compatible with nitrates and lower (acidic) pH. All metallic nitrates are inorganic salts of a given metal cation and the nitrate anion.