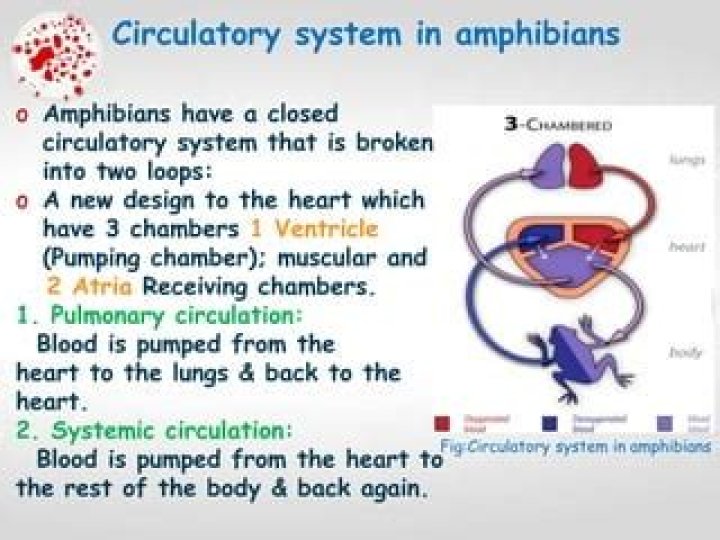
How many electrons will be shown on the molecular orbital diagram for o2

Andrew Mccoy
Updated on April 08, 2026
Each O atom contributes 8 electrons to the O₂ molecule. You use the Pauli Exclusion Principle and Hund’s Rule to add the 16 electrons to the molecular orbitals in an Aufbau process.
How many molecular orbitals are formed in O2?
Therefore, there will be eight molecular orbitals in O2. Each oxygen bonds to the other with its 1s, 2s, and 2p orbitals. these 5 atomic orbitals combine to form 10 molecular orbitals.
How many orbitals are in oxygen?
The valence orbitals in an isolated oxygen atom are a 2s orbital and three 2p orbitals. The valence orbitals in an oxygen atom in a water molecule differ; they consist of four equivalent hybrid orbitals that point approximately toward the corners of a tetrahedron (Figure 2).
How many bonding electrons are shown in the O2 molecule pictured in the diagram above?
They are shown in red in the above diagram. This gives you a total of 6 anti-bonding electrons. This tells you that the two oxygen atoms are bonded together via a double bond, which of course consists of a sigma and a pi bond.How many inner shell electrons does oxygen have?
That means there are 8 electrons in an oxygen atom. Looking at the picture, you can see there are two electrons in shell one and six in shell two.
When O2 changes to O2 the electron goes to which of the orbitals?
The electrons are filled from the orbital of lowest energy to the orbital of highest energy. So, the correct answer is “Option C”.
What bond order does O2 2 have?
The bond order for the O22- is 1 (one). In a neutral O2 molecule, there are a total of 12 valence shell electrons shared between the bonded atoms.
What is the bond order of C2?
The bond order of C2 molecule is 2.How do you calculate bond order o2?
The bond order may be defined as half the difference between the number of electrons in bonding molecular orbitals (Nonbonding) and the number of electrons in the antibonding molecular orbital. The bond order is the number of bonds present between two atoms in a molecule or ion.
How do I calculate bond order?- Draw the Lewis structure.
- Count the total number of bonds.
- Count the number of bond groups between individual atoms.
- Divide the number of bonds between atoms by the total number of bond groups in the molecule.
How many protons and electrons are in oxygen?
Answer 2: Oxygen is the 8th element in the periodic table. This means that oxygen has 8 protons and 8 electrons. In order to get the number of neutrons you take the atomic weight in this case 15.9999~16 and you subtract it by the number of protons (16-8).
What is oxygen electron?
Oxygen is the eighth element with a total of 8 electrons. In writing the electron configuration for oxygen the first two electrons will go in the 1s orbital. Since 1s can only hold two electrons the next 2 electrons for O go in the 2s orbital. … Therefore the O electron configuration will be 1s22s22p4.
How many neutrons does O2 have?
GeneralSymbol16ONamesoxygen-16, O-16, Oxygen – 16Protons8Neutrons8
How many shells does oxygen have?
ZElementNo. of electrons/shell5Boron2, 36Carbon2, 47Nitrogen2, 58Oxygen2, 6
How many electrons does oxygen have in its outer shell?
Valence electrons are the electrons in the outermost shell, or energy level, of an atom. For example, oxygen has six valence electrons, two in the 2s subshell and four in the 2p subshell. We can write the configuration of oxygen’s valence electrons as 2s²2p⁴.
Can oxygen have 7 electrons?
Carbon has four valence electrons, and oxygen has six. … When this is done, each oxygen atom has a total of seven valence electrons and the carbon atom has a total of six valence electrons. Because none of these atoms have an octet of valence electrons, we combine another electron on each atom to form two more bonds.
What is the bond length of O2?
Molecular FormulaNameBond Length (Å)O2oxygenO-O 1.208[O2]+O-O 1.1227[O2]-(s)O-O 1.28 (s=0.01)[O2]2-(s)O-O 1.49 (s=0.009)
Does SP mixing occur in N2?
N2 has s-p mixing, so the π orbitals are the last filled in N2+2. N 2 2+ . O2 does not have s-p mixing, so the σp orbital fills before the π orbitals.
How many electrons occupy the antibonding molecular orbital for the N2 molecule?
There are five antibonding orbitals in a molecule of N₂.
Which of the following is paramagnetic a O2 B CN C CO D NO+?
Explanation: CN- , CO and NO+ are isoelectronic with 14 electrons each and there is no unpaired electrons in the MO configuration of these species. So these are diamagnetic. O-2 is paramagnetic due to the presence of one unpaired electron.
Is O2 2 paramagnetic or diamagnetic?
O2, O2^ – are paramagnetic while O3, O2^2 – are diamagnetic.
How many electrons does C2 have?
So it has 6 electrons and protons while it’s electronic configuration is 1s2 ,2s2,2p2. In C2 , two atoms of carbon bonded together to form a molecule.
What is the bond order of N2+ N 2?
That is, the bond order for N2+ is 2.5.
How do you find bonding electrons?
- 6 (S)+2⋅6 (2O)=18.
- 8 (S)+2⋅8 (2O)=24.
- 24−18=6, i.e. three bonding electron pairs.
- 18−6=12 i.e. six lone pairs.
Why is 02 paramagnetic?
Based on molecular orbital theory, orbitals are formed by overlapping atomic orbitals of oxygen atoms. … Due to the presence of two unpaired electrons, we can say that the oxygen molecule is paramagnetic in nature. The reason why oxygen is paramagnetic is because of the presence of two unpaired electrons.
How many neutrons and electrons are in oxygen?
Oxygen has eight protons, eight neutrons, and eight electrons. Oxygen is different from carbon because it has more particles. Each kind of atom has a different number of the three particles. Protons and neutrons are in the center, or nucleus, of the atom.
How many neutrons are in oxygen 15?
Oxygen-15 is a radioactive isotope of oxygen, frequently used in positron emission tomography (PET) imaging. It can be used, amongst other applications, in water for PET myocardial perfusion imaging and for brain imaging. It has 8 protons, 7 neutrons, and 8 electrons. The total atomic mass is 15.0030654 amu.